First Principles Simulation of Glycine Adsorption to Amorphous Silica
- Started
- 27th September 2012
- Ended
- 8th May 2013
- Research Team
- Benjamin Lowe
- Investigators
- Chris-Kriton Skylaris
This work was performed as part of a Independent Research Project as part of the MSc component of the Complex Systems Simulation Doctoral Training Center Course. The aim of the project was to learn about Quantum Chemistry Simulation in the context of an area I am interested in - Silica/Biomolecule interaces.
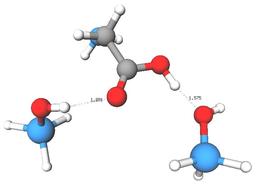
The interface between biological molecules and inorganic surfaces is currently poorly understood at a molecular scale due to its inherent complexity, and of great importance in areas such as Bionanotechnology. Replicating the work of Lomenech et al. [1], the adsorption of neutral glycine onto amorphous silica was investigated using Density Functional Theory (DFT) calculations at the B3LYP/6-31++G** level using a cluster-based approach. Various binding configurations were geometry optimised, and their vibrational frequencies calculated and compared to experimental data. With one silanol, the most favorable binding mode was via the carboxyl group acting as both a H-bond donor and acceptor. With two silanols this was supplemented with additional H-bonds from the amine moiety, resulting in a increased adsorption energy. Vibrational calculations demonstrate a strong bathochromatic shift of the carbonyl stretch with increasing numbers of H-bonds between the carboxyl and the silanols which, supported by IR-spectroscopy data, suggests that several H-bonds occur between the glycine and silica surface.
[1] C. Lomenech, G. Bery, D. Costa, L. Stievano, and J. F. Lambert, “Theoretical and experimental study of the adsorption of neutral glycine on silica from the gas phase,” vol. 6, no. 6, p. 10611070, 2005
Categories
Life sciences simulation: Biomolecular simulations
Physical Systems and Engineering simulation: Materials
Algorithms and computational methods: Quantum Chemistry
Simulation software: NWCHEM
Visualisation and data handling software: ECCE
Computational platforms: Linux
Transdisciplinary tags: Scientific Computing