Multiscale modelling of biological membranes
- Homepage
- http://www.personal.soton.ac.uk/orsi/researchMario.html
- Started
- 12th November 2009
- Ended
- 11th November 2012
- Research Team
- Mario Orsi
- Investigators
- Jonathan Essex
Biological membranes are fundamental structures employed by nature to encapsulate cells; they are dynamic systems in which proteins float in a sea of lipids. Biomembranes and associated proteins are fundamental components of many phenomena indispensable for life, such as growth, energy storage, and in general information transduction via neural activity. Lipid bilayers, which constitute the backbone structure of any biological membrane, also play increasingly crucial roles in biomedical technology, particularly in drug research and development.
A general problem in this field is that experimental evidence is difficult to obtain and be rationalised, due to the small scale and complexity of the systems studied. Computer simulation is an established methodology which can be used to help in the interpretation of experimental data, guide new lines of investigation and ultimately understand biomembrane structures and phenomena with molecular-level resolution. However, standard simulation techniques are computationally expensive, often prohibitively so. To overcome this problem, in recent years much research has been focused on simplified, so-called "coarse-grain" models; coarse-grain simulations are faster but less accurate than standard simulations.
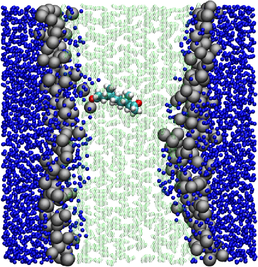
In this project we wish to develop further a "multiresolution" methodology, where efficiency is optimised by modelling the bilayer environment at the coarse-grain level, while accuracy is maintained by retaining a standard atomic-level description of membrane-associated proteins and drugs. Preliminary results obtained with this method are very promising, but substantial effort is required to extend it towards applications to the most fundamental yet complex problems of membrane biophysics. The technique would be fast and accurate enough to be capable of shedding light on the currently much debated roles that the interactions between lipids and proteins play in membrane biology. We also propose to employ the techniques developed to areas of biomedical interest, particularly drug design. All drugs must permeate the cell membrane, and most of them target membrane proteins. The mechanisms of drug permeation and action are difficult to study by experiments or standard simulations; we will apply the multiscale methodology to better understand such phenomena and hence guide the design of improved or novel drugs. Moreover, we will investigate problems relevant in nanotechnology. In particular, this work could provide quantitative insights into the controversial issues regarding the interactions between cell membranes and nanomaterials. Such work would be relevant for the promising biomedical application of nanomaterials, for instance as drug delivery agents. We will also use our methodology to assess the important issue of toxicity and biocompatibility of nanomaterials.
Categories
Life sciences simulation: Biomedical, Biomolecular Organisation, Structural biology
Algorithms and computational methods: Molecular Dynamics, Multi-scale
Visualisation and data handling software: Xmgrace
Software Engineering Tools: Emacs, SVN